Insights+: The US FDA New Drug Approvals in February 2023
Shots:
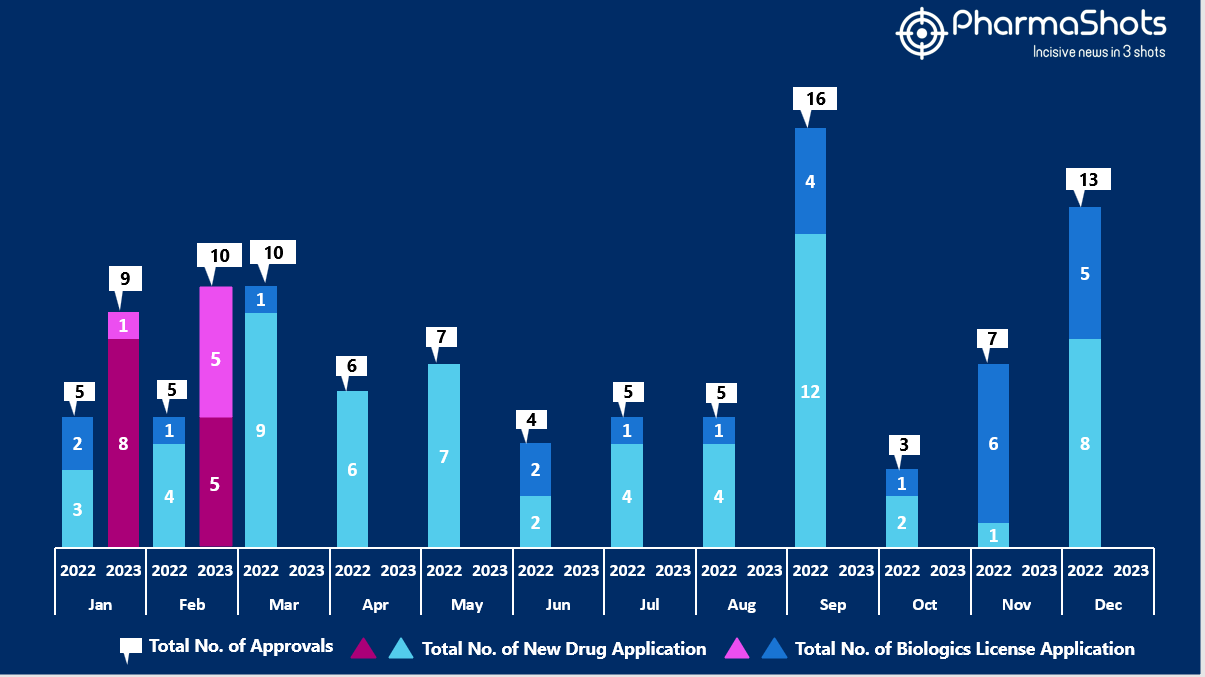
- The US FDA approved 5 NDAs and 5 BLA in February 2023, leading to treatments for patients and advances in the healthcare industry. The CDER and CBER approved 19 novel products in 2023
- In February 2023, the major highlights drugs were, Tezspire (tezepelumab) approval for severe asthma, Altuviiio for hemophilia A
- PharmaShots has compiled a list of a total of 5 new drugs approved by the US FDA in February 2023
GSK’s Jesduvroq (daprodustat) Receives the US FDA’s Approval for the Treatment of Anaemia
Jesduvroq
Active ingredient: daprodustat Approved: February 02, 2023
Company: GSK Disease: Anaemia
- The US FDA has approved Jesduvroq, an oral hypoxia-inducible factor prolyl hydroxylase inhibitor as 1st oral treatment of anaemia due to CKD undergoing dialysis
- The approval was based on the P-III (ASCEND-D) trial results evaluating Jesduvroq vs recombinant human erythropoietin in 2964 dialysis patients who were switched to receive daprodustat ESA control from an SoC ESA therapy
- The results showed that daprodustat was non-inferior to ESAs regarding the change in the Hb level from baseline & CV outcomes, the major adverse cardiovascular event (25.2% vs 26.7%) during a median follow-up of 2.5yrs., patients with other AEs were similar in 2 groups, mean change in Hb level from baseline to 28-52wks. The MAA for daprodustat is under EMA’s review with a regulatory decision expected in H1’23
Tezspire
Active ingredient: tezepelumab Approved: February 03, 2023
Company: AstraZeneca and Amgen Disease: Asthma
- The US FDA has approved Tezspire for self-administration in a pre-filled, single-use pen for patients aged ≥12yrs. with sev. asthma
- The approval was based on the (PATHFINDER) program incl. results from the P-I trial (PATH-BRIDGE) & P-III trial (PATH-HOME) evaluating Tezspire (210mg). In the (PATH-HOME) trial, 92% of healthcare providers, patients, and caregivers were able to successfully administer Tezspire in the clinic & home along with an improvement in asthma control & safety profile was consistent with prior trials
- Tezspire is also supplied in a single-dose vial or prefilled syringe. The therapy was approved in the US, EU, Japan & other countries and is under regulatory review in multiple other countries globally
Eylea
Active ingredient: aflibercept Approved: February 09, 2023
Company: Regeneron Disease: Retinopathy
- The US FDA has approved Eylea (VEGF inhibitor) to treat preterm infants with ROP. The approval was based on the P-III trial (FIREFLEYE & BUTTERFLEYE) evaluating Eylea (0.4mg) vs laser photocoagulation in 113 & 120 patients
- Eylea is being jointly developed by Regeneron & Bayer. Both trial results showed that 80% achieved an absence of active ROP & unfavorable structural outcomes @52wks. with no new safety signals
- In (FIREFLEYE & BUTTERFLEYE) trials, ocular AEs (39% vs 37%) & (18% vs 26%); SAEs (8% for both groups) & (6.5% vs 11%), AEs were consistent with infant prematurity or to the inj. procedure. The (FIREFLEYE) results were published in JAMA while (BUTTERFLEYE) results were presented at ROP update 2022
Jemperli
Active ingredient: dostarlimab-gxly Approved: February 10, 2023
Company: GSK Disease: Endometrial Cancer
- The US FDA has granted full approval for Jemperli in recurrent or advanced dMMR advanced endometrial cancer that has progressed on or after a prior Pt-containing regimen in any setting and are not a candidate for curative surgery or radiation.
- The approval was based on additional data from the A1 expansion cohort of an ongoing P-I trial (GARNET) evaluating Jemperli monotx. Patients in cohort A1 received Jemperli (500mg, IV, q3w) for 4 doses, followed by 1000mg, q6w
- The results showed ORR (45.4%) with CR rate (15.6%) & PR rate (29.8%), m-DoR was not reached, and patients had a duration of response ≥1 & ≥2yrs. (85.9% & 54.7%), median follow-up for DoR was 27.9mos., TRAEs were consistent with prior analyses for cohort A1
Lamzede
Active ingredient: velmanase alfa-tycv Approved: February 16, 2023
Company: Chiesi Disease: Alpha-Mannosidosis
- The US FDA has approved Lamzede, a recombinant human alpha-mannosidase for adult & pediatric patients with non-central nervous system manifestations of alpha-mannosidosis. The approval was based on multiple trials incl. the P-III & P-II trial evaluating velmanase alfa (1mg/kg, qw, IV) vs PBO
- In the P-III trial, velmanase alfa showed an improvement in the 3-minute stair climbing test, 6MWT & forced vital capacity @12mos. & efficacy was supported by a reduction in serum oligosaccharide concentration. The P-II trial showed the mean absolute & percentage changes from baseline for serum oligosaccharides
- Lamzede received marketing authorization from the EC in 2018 for non-neurological manifestations in patients with mild to mod. AM
Austedo XR
Active ingredient: deutetrabenazine Approved: February 17, 2023
Company: Teva Pharmaceutical Disease: chorea
- The US FDA has approved Austedo XR (vesicular monoamine transporter 2 inhibitors), a new qd formulation of deutetrabenazine for adults with tardive dyskinesia (TD) and chorea associated with Huntington’s disease. The product is expected to be available in the US in 2023
- When switching b/w Austedo (BID) tablets and Austedo XR (qd) tablets, the qd formulation of Austedo XR was therapeutically equivalent to the BID formulation.
- Austedo XR is supplied as 6mg, 12mg, and 24mg strength extended-release tablets and can be taken with/without food. Additionally, the product was approved in the US for adults for the same indication
Apellis’ Syfovre Receives the US FDA’s Approval as the First Treatment for Geographic Atrophy
Syfovre
Active ingredient: pegcetacoplan Approved: February 21, 2023
Company: Apellis Disease: Geographic Atrophy
- The US FDA has approved Syfovre (pegcetacoplan injection) for GA secondary to AMD. The approval was based on the P-III (OAKS) & (DERBY) studies evaluating the efficacy & safety of Syfovre
- The results showed a reduction in the rate of GA lesion growth vs sham and demonstrated increasing treatment effects over time with the greatest benefit, ~36% reduction in lesion growth with monthly treatment in (DERBY) b/w 18-24mos.
- The company launched the patient support program i.e., ApellisAssist incl. insurance support, financial assistance, disease education & ongoing product support for eligible patients. The MAA for Syfovre is currently under EMA’s review with an expected decision in early 2024
Filspari
Active ingredient: sparsentan Approved: February 21, 2023
Company: Travere Therapeutics Disease: IgA Nephropathy
- The US FDA has granted accelerated approval to Filspari to reduce proteinuria in adults with primary IgAN who are at risk of rapid disease progression. Filspari is expected to be available in Feb 2023
- The approval was based on an ongoing (PROTECT) study evaluating sparsentan (400mg) vs irbesartan (300mg) in 404 patients aged ≥18yrs. which showed a significant improvement in proteinuria & the therapy was well tolerated with a consistent safety profile across all clinical trials
- The 2yr. results from the confirmatory EPs analysis are expected in Q4’23 & will support traditional approval of Filspari. The company has launched a patient support program Travere TotalCare incl. service, assistance & resources for eligible patients
Sanofi’s Altuviiio Receives the US FDA’s Approval for the Treatment of Hemophilia A
Altuviiio
Active ingredient: efanesoctocog alfa Approved: February 24, 2023
Company: Sanofi Disease: Hemophilia A
- The US FDA has approved Altuviiio indicated for routine prophylaxis & on-demand treatment to control bleeding episodes & perioperative management for adults & children with hemophilia A. The product will be available in April in the US
- The approval was based on the P-III study (XTEND-1) evaluating Altuviiio (qw) in 159 patients aged ≥12yrs. The trial met its 1EPs & 2EPs i.e., significant bleed protection with a mean ABR of 0.70, median ABR of 0.0, 77% reduction in ABR vs prior factor prophylaxis
- Prevention of joint bleeds with a median annualized joint bleeding rate of 0, 100% resolution of target joints, mean factor VIII activity ≥40% for most of the week & ≥10% at 7 Days, was well-tolerated with no reports of factor VIII inhibitor development. Regulatory submission of Altuviiio in the EU is expected in H2’23
Skyclarys
Active ingredient: omaveloxolone Approved: February 28, 2023
Company: Reata Pharmaceuticals Disease: Friedreich’s Ataxia
- The US FDA has approved Skyclarys for adults & adolescents aged ≥16yrs. with Friedreich’s ataxia. The approval was based on the (MOXIe) part 2 trial & post hoc propensity-matched analysis of the OLE (MOXIe) trial evaluating omaveloxolone (150mg) vs PBO
- The (MOXIe) part 2 trial results showed lower mFARS scores over PBO @48wk., the PBO-corrected difference b/w the 2 groups were -2.41 points. In the OLE trial, improved mFARS scores after 3yrs. over untreated patients from FA-COMS natural history study
- The company launched a patient services program & provides access to patients for Skyclarys treatment. The product is expected to be available in Q2’23 and received ODD, FTD & RPDD from the US FDA; ODD from the EC for Friedreich’s ataxia
Related Post: Insights+: The US FDA New Drug Approvals in January 2023
Tags

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.