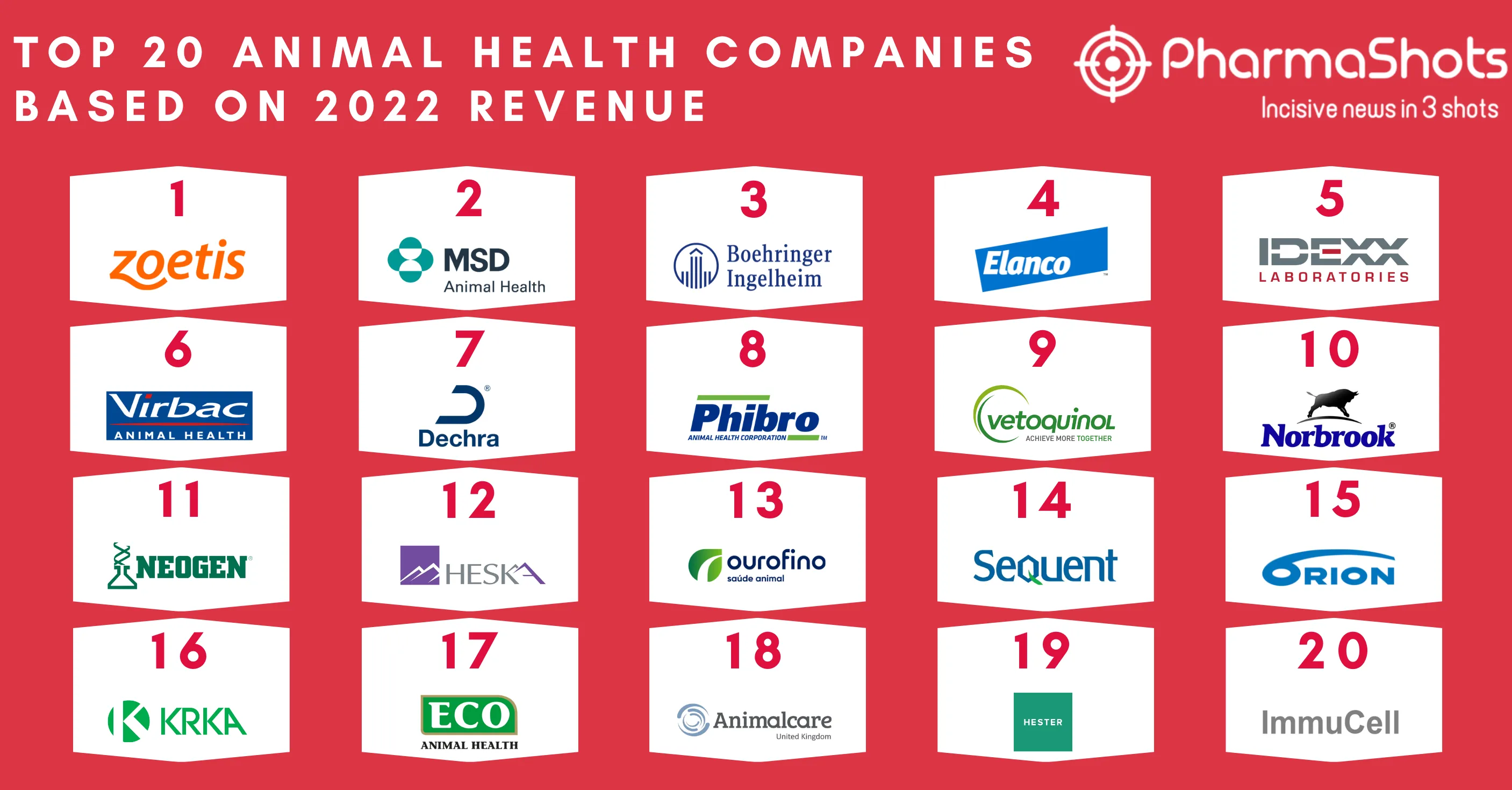
Zoetis’ Librela (bedinvetmab) Receives the US FDA’s Approval to Control Osteoarthritis Pain in Dogs
Shots:
- The US FDA has approved the first anti-NGF mAb treatment, Librela (qm) for the control of pain associated with OA in dogs
- The results from 2 field studies showed a reduction in OA over PBO and reduces pain. Acc. to studies, Librela improves people's mobility & overall QoL while Librela's effectiveness might not become apparent until after 2nd dose, dogs may also experience a reduction in pain as early as 7days after 1st dose
- Additionally, dogs treated with bedinvetmab experienced lasting OA pain relief over the course of the study with monthly inj. in a continuation study. The therapy is expected to be available in the US in late 2023 & was approved for use in Canada, Brazil, Australia, New Zealand, Japan & other markets
Ref: Zoetis | Image: Zoetis
PharmaShots! Your go-to media platform for customized news ranging for multiple indications. For more information connect with us at connect@pharmashots.com
Click here to read the full press release

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.