Most Viewed Articles of 2023
2023, remained a busy year for PharmaShots. To keep our readers up to date on the developments in the healthcare sector, PharmaShots worked steadfastly to deliver real-time and real-world updates. Here’s a synopsis of our key reports and most-read news of the year.

Top 20 Radiopharma Companies Based on Market Cap
Shots:
- Integrating scintigraphy with therapeutics, radiopharmaceutical companies have emerged profoundly in the past few decades. With the growing influence of radiopharmaceutical companies in oncology, the companies hold a firm grasp on the market
- With the expanding acceptance among HCPs, patients, and researchers, radiopharmaceutical companies are estimated to touch the $7.5B threshold by 2026. This year, Novartis has secured the top position with a market cap of $207.65B, followed by Bayer and Lantheus Medical Imaging
- PharmaShots has compiled a list of the Top 20 Radiopharma Companies developing products as a treatment option, ranked based on their market cap
Top 20 IPOs in Healthcare by Total Fund Value
Shots:
- Immutable capital demands and high-spending drug development plans are the most common drivers in encouraging fully-fledged private healthcare companies to go public. Apart from the exposure and capital raise, companies gain more recognition, credibility, and indispensable value assessment
- 2022 had been a great year when it comes to the IPOs of healthcare companies. Bausch + Lomb efficaciously emerged as a top-grade performer with a total IPO value of $711M, followed by HilleVax and Third Harmonic Bio
- With the invaluable insights of DealForma, PharmaShots has prepared a detailed report on the Top 20 IPOs in the Healthcare industry of the year 2022
Top 20 Cell and Gene Therapy Companies Based on 2022 Total Revenue
Shots:
- Standing tall and gradually paving its way through extensive research, Cell and Gene Therapy is a beacon of hope when treating rare and inherited diseases. Cell and gene therapy aims to prevent, treat, and potentially reduce the effects of the underlying cause of genetic diseases.
- Propelled by constant innovatory winds, the Cell and Gene therapy market is anticipated to grow by 20 percent year-over-year through 2025. With a total revenue of $52B, Merck & Co. Was ranked the highest among the list of companies developing Cell & Gene therapy, followed by Novartis with a revenue of $50.54B and Spark Therapeutics with a revenue of $49.27B
- PharmaShots brings an informative report on the Top 20 Cell and Gene Therapy Companies based on the total revenue generated in the year 2022
Top 20 Medical Device Companies Based on 2022 Total Revenue
Shots:
- Driven by constant innovation and life-changing technological advances, medical device companies have integrated a holistic approach to diagnosis and therapeutics. Bolstered by the inch-perfect result and cutting-edge technology, gradually medical devices have got profoundly embedded in our lives
- With a global market value of $562.6B in 2022, medical device companies are anticipated to touch the astronomical threshold of $965.2B. Medtronic led the medical device industry this year with a total sale of $30.11 billion, followed by Johnson & Johnson and EssilorLuxottica.
- PharmaShots brings this month an informative report on the Top 20 Medical Device Companies Based on 2022 Revenue
Top 20 Drugs with US Patent Expiry in 2023 Based on Total Sales Value
Shots:
- Drug patent expiry is when a patent granted to a pharmaceutical company for a particular drug expires, allowing other companies to produce and sell generic versions
- Like every other utility patent, pharmaceuticals also get market exclusivity of 20 years. Some companies accept the offered period and open doors to biological drugs or biosimilars. In contrast, others try different strategies like new formulations, developing alternative administration techniques, and modifying the drug uses to extend the market exclusivity
- While several other factors, like the unanticipated arrival of a better drug, failure to disburse maintenance fees, and post-M&A restructured pipelines, can be early instigators of patent expiry. PharmaShots has prepared a list of the top 20 drugs with patent expiry in 2023

Insights+: Key Takeaways of J.P. Morgan Healthcare Conference 2023 Based on Therapeutic Areas
Shots:
- The US FDA approved 8 NDAs and 5 BLA in December 2022, leading to treatments for patients and advances in the healthcare industry. The CDER and CBER approved 86 novel products in 2022
- In December 2022, the major highlights drugs were Krazati (adagrasib) approval for non-small cell lung cancer with a KRASG12C mutation, Sunlenca (lenacapavir) for multi-drug resistant HIV
- PharmaShots has compiled a list of a total of 13 new drugs approved by the US FDA in December 2022
Insights+: The US FDA New Drug Approvals in April 2023
Shots:
- The US FDA approved 6 NDAs and 4 BLA in April 2023, leading to treatments for patients and advances in the healthcare industry. The CDER and CBER approved 36 novel products in 2023
- In April 2023, the major highlights drugs were, Trikafta approval for children with cystic fibrosis, Qalsody for the treatment of amyotrophic lateral sclerosis
- PharmaShots has compiled a list of a total of 10 new drugs approved by the US FDA in April 2023
Top Performing Drug – Dupixent (April Edition)
Shots:
- In continuation of our previous series on the top-performing drug of the month, based on 2021 revenue, this month we have selected Dupixent and prepared a curated analysis report for our readers
- Dupixent is an interleukin-4 receptor alpha antagonist used for the treatment of multiple indications including atopic dermatitis, asthma, eosinophilic esophagitis, etc.
- PharmaShots presents an informative take on the key features of Dupixent with a detailed analysis of its revenue, clinical trials, alternatives, and approvals. The report is concluded with an engaging SWOT analysis and edifying KOL reviews
JP Morgan Special: Dealmaker 2022 – Top M&A Deal of Amgen (Part 02)
Shots:
- In the year 2022, Amgen was the top dealmaker, based on the total deal value, with 6 deals at a total announced value of ~$34.5B. Amgen has disclosed 5 out of the 6 deals leaving its BigHat Biosciences deal undisclosed
- Amgen’s acquisition of Horizon Therapeutics for a value of ~$28.3B was the key contributor towards the total deal value gained by Amgen in the year 2022
- Amgen was followed by Pfizer, Sanofi, and J&J among the top deal makers of the year. Our team at PharmaShots has compiled an insightful report by utilizing the data collected by DealForma for the Deal Maker of the Year 2022
Top Performing Drug of 2021 – Trulicity (February Edition)
Shots:
- In continuation of our previous series on the top-performing drug of the month, based on 2021 revenue, this month we have selected Trulicity and prepared a curated analysis report for our readers
- Trulicity is a glucagon-like peptide-1 (GLP-1) receptor agonist used for the treatment of type 2 diabetes and to reduce the risk of major adverse cardiovascular events in adult patients with type 2 diabetes and established cardiovascular disease or multiple cardiovascular risk factors
- PharmaShots presents an informative take on the key features of Trulicity with a detailed analysis of its revenue, clinical trials, alternatives, and approvals. The report is concluded with an engaging SWOT analysis and edifying KOL reviews
Insights+ Key Biosimilars Events of January 2023
Shots:
- Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency
- Biosimilars are expected to be a cost-effective alternative to the high-priced branded biologics, offering significant and much-needed cost savings to both payers and the patients
- During the month of January, Celltrion reported P-III trial results of CT-P16 (biosimilar, bevacizumab) for non-squamous non–small cell lung cancer, STADA and Xbrane received the MHRA’s approval for Ximluci (biosimilar, ranibizumab) to treat retinal diseases. Our team at PharmaShots has summarized 16 key events of the biosimilar space of January 2023
JP Morgan Special: Dealmaker 2022 – Top M&A Deal of Roche (Part 01)
Shots:
- Roche was the top dealmaker in 2022 with a completion of 31 deals with multiple pharma, biotech companies, and universities for a total deal value of $13.55B
- The highest deal was secured by Roche under its development and commercialization collaboration with Poseida for the R&D of multiple existing and novel cell therapies for the treatment of multiple myeloma, B-cell lymphomas, and other hematologic indications
- Sanofi, Eli Lilly, and BMS are also among the top dealmakers. Leveraging the data from DealForma, our team at PharmaShots has compiled insightful data for the Deal Maker of the Year
Insights+: The US FDA New Drug Approvals in January 2023
Shots:
- The US FDA approved 8 NDAs and 1 BLA in January 2023, leading to treatments for patients and advances in the healthcare industry. The CDER and CBER approved 9 novel products in 2023
- In January 2023, the major highlights drugs were Rykindo (risperidone) approval for schizophrenia and bipolar 1 disorder, Brenzavvy (bexagliflozin) for type 2 diabetes
- PharmaShots has compiled a list of a total of 9 new drugs approved by the US FDA in January 2023
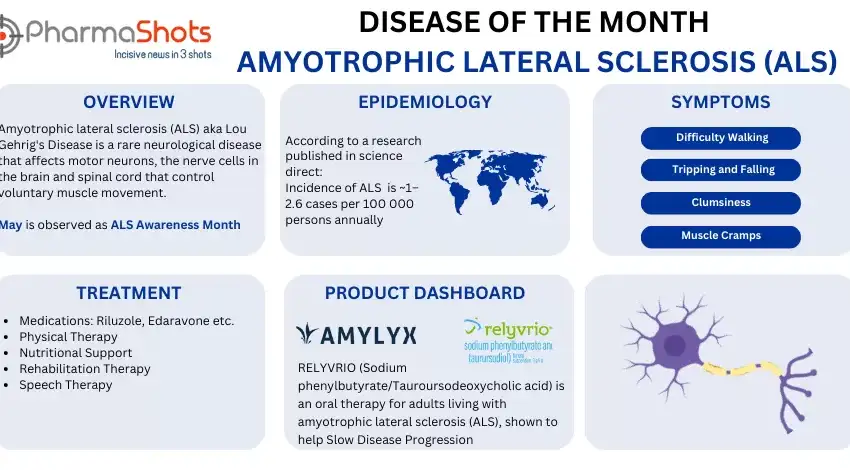
Disease of the Month: Hemophilia
Shots:
- To create a wholesome environment for the current and the future generation, it’s totally up to life science companies and decision-makers to embrace and integrate every prospect of the underlying disease conditions and address those issues to find cures
- To continue the series for disease of the month, PharmaShots brings this month a summary of the disease Hemophilia, a rare, genetic disorder that slows down the blood clotting process
- 17th April is observed as World Hemophilia Day to raise awareness for hemophilia and other bleeding disorders.
Top Performing Drug – Trikafta/Kaftrio (May Edition)
Shots:
- In continuation of our previous series on the top-performing drug of the month, based on 2021 revenue, this month we have covered Trikafta/Kaftrio as the top-performing drug of the month
- Trikafta is a combination of ivacaftor, tezacaftor, and elexacaftor indicated for the treatment of cystic fibrosis in pediatric patients who have at least one F508del mutation in the CFTR gene
- PharmaShots presents an informative take on the key features of Trikafta/Kaftrio with a detailed analysis of its revenue, clinical trials, alternatives, and approvals. The report is concluded with an engaging SWOT analysis and edifying KOL reviews

6 Key Factors Influencing the Pharmaceutical Industry
Shots:
- With several swaying factors exerting a direct or indirect impact on the overall functioning of the pharmaceutical industry, understanding the nitty-gritty of the operational model of the industry can be clumsy
- PharmaShots presents an intuitive article on 6 key factors that exercise control over the pharmaceutical industry
- Sit back and relax, while the article unfolds before you, the magic model that is certain to revamp your company
8 Major Reasons Behind the Soaring Layoffs in the Pharmaceutical Industry
Shots:
- In the wake of International Labour Day, PharmaShots presents a detailed take on the recent layoffs in the Pharmaceutical Sector with an illustrative graphical analysis
- Like any sector, the pharmaceutical industry operates on some nuance and intricate aspects that ensure smooth operations. Factors like digitalization, patent expiry, growing competition, and others put direct or indirect impacts on the revenue of the companies
- Ever wondered, why for the past few days our feed has been brimming with layoffs news? The pharmaceutical industry is an ever-growing sector driven by innovation and astounding technology. Check out our intuitive article on “8 Major Reasons Behind the Soaring Layoffs in the Pharmaceutical Industry

Shots:
- Anne started by talking about the launch of the first-ever cleft surgery virtual reality platform in collaboration with Biodigital, which simulates surgery on cleft lip and palate, a birth condition
- Anne explained how the VSS VR Extension technology enables surgeons worldwide to collaborate in virtual operating rooms, ensuring safer surgery and better outcomes for patients with clefts
- The interview showcases how Smile Train supports local medical professionals with training, funding, and resources to provide free cleft surgery and comprehensive cleft care to children globally
Shots:
- Iris shared the specifics of the launch of the UCB’s Innovators campaign. The campaign spotlights the R&D employees’ perspectives on innovation and aims to highlight why innovation is a key driving force at UCB for bettering the lives of patients
- She gave her thoughts on how UCB is adopting innovative AI programs that use advanced analytics and algorithms to generate actionable outcomes from patient insights
- Through this interview, UCB’s focus on continuous innovation across all dimensions of research and development to solve the challenges in the lives of people living with severe diseases can be understood
Shots:
- Stacey talked about the launch of its first streaming commercial as part of a direct-to-consumer advertising campaign for adults with plaque psoriasis
- The “Topical Uprising” campaign focuses on the patient journey and taps into the frustrations & self-conscious of living with plaque psoriasis that helps patients to demand more from their topical plaque psoriasis treatment
- The interview shows how Dermavant is promoting advertising campaigns to commercialize its innovative therapeutics in immuno-dermatology
Johanna Contreras and Lupe Share Insights from the Cuide Su Corazón Campaign
Shots:
- Johanna Contreras, Director of the Ambulatory Heart Failure Network & Diversity Mount Sinai Health System spoke about the initiation of this campaign and how it is empowering Latina women with heart failure to prioritize their heart health
- Lupe, a woman diagnosed with heart failure, shared her experience of her life with heart failure and how this campaign helped her in the improvement of her condition
- Cuide Su Corazón is the second phase of the Hear Your Heart campaign launched in April 2022 by Boehringer & Lilly. The program was built to deepen the connection with Latina women living with heart failure in Spanish-language content and resources to support these women
Insights from Coralie Gressier-Sayag on Avapritinib Efficacy Data Presented at EHA 2023
Shots:
- Coralie started by giving a brief introduction about Systemic Mastocytosis (SM), a rare disease. She told us that in Europe, approximately 40,000 people live with SM, with 80-90% living with Indolent SM and the rest with Advanced SM
- She then gave an overview of the P-II PATHFINDER study evaluating avapritinib. The study’s 2-year follow-up data validates avapritinib’s role in treating Advanced SM, providing long-lasting benefits and robust responses
- The interview shows how Blueprint Medicines remains committed to research and development, as well as educational initiatives for patients and healthcare providers, underlining their dedication to transforming care for rare diseases like SM

Shots:
- Dr. Philip shared his views on the collaboration of Sernova with Evotec for iPSC-Based Beta cell replacement therapy to develop and commercialize a ‘Functional Cure’ for diabetes.
- He also gave an overview on the benefits of Evotec’s iPSC-based insulin-producing beta cells on the next generation of Sernova’s Cell Pouch System
- The interview discusses about how Sernova is developing regenerative medicine therapeutic technologies to improve the treatment and quality of life of people with chronic metabolic diseases
Shots:
- Dr. Richard talked about the results from the NefIgArd P-III trial evaluating Tarpeyo (Nefecon) for the treatment of IgA Nephropathy
- He also highlighted the safety and efficacy results from the NefIgArd study. He also spoke more about Trapeyo and the roadmap for its global approval and commercialization
- The interview shows up how Calliditas is developing novel treatments in orphan indications, in addition to renal and hepatic diseases
Shots:
- Laura gave an overview of Human acellular vessels (HAV). She talked about the six-month results of the HAV in a non-human primate model of CABG presented at the annual American Heart Association (AHA) Scientific Sessions 2022
- She also elaborated on the construction and development of the CABG model and how it treats blocked arteries in the heart
- The interview summarizes how Humacyte’s HAV can transform coronary artery bypass grafting providing greater durability of surgical results and decreased prevalence
Shots:
- Jeff spoke about the first patient enrolled in a P-II study of D-4517.2 for the treatment of wet AMD and DME
- Jeff also talked about Ashvatta’s Hydroxyl Dendrimer Platform and how this technology is beneficial to treat AMD and DME
- The interview gives a profound understanding of how Ashvattha is developing hydroxyl dendrimer therapeutics to target unmet medical needs in ophthalmology, neurology, inflammation, and neuro-oncology
Mark Cobbold and Carsten Linnemann Share Insights on Neogene Therapeutics’ Acquisition
Shots:
- Carsten gave the details of the agreement with Neogene Therapeutics to bring cell therapies to patients with solid tumors through Neogene’s next-generation T-cell receptor therapies. He also talked about Neogene’s TCR discovery platform
- Mark spoke about how this acquisition provides AstraZeneca with a unique opportunity to bring innovative science and leading experts in T-cell receptor biology and cell therapy manufacturing to find new ways to target cancer
- Under the agreement, AstraZeneca acquired all outstanding equity of Neogene Therapeutics for a total consideration of up to $320m, on a cash and debt-free basis. The transaction closed in the first quarter of 2023
Shots:
- Toni spoke about the CONNEQT Pulse the first dual blood pressure monitor with ATCOR’s SphygmoCor technology. The device is used for remote patient monitoring, home health, and clinical trials
- She also elaborated about the data used for the pre-marketing submission with FDA. She explained the seven key vascular biomarkers that the device measures and data validating its accuracy
- The interview gives an understanding of how CardieX along with its subsidiaries ATCOR and CONNEQT are working to advance innovative medical devices, digital solutions, and wearables to raise the standard of care
Shots:
- Andrea talked about the launch of a digital patient support resource by the Ovarian Cancer Commitment (OCC) in co-development with AstraZeneca and other organizations for ovarian cancer patients
- She also highlighted how this important resource, named Olivia supports in navigating ovarian cancer diagnosis. This tool is an interactive tool that allows users to access the content they need at the time they need it, to customize their learning experience
- The interview showcases the expanding collaborations between different organizations to deliver comprehensive information with the goal of improving the outcomes and quality of life of patients with ovarian cancer
Shots:
- Nirja gave the details of the PATHWAY anti-HER2 (4B5) to identify metastatic breast cancer patients with low HER2 expression through its scoring algorithm that helps pathologists to identify “low expressors” of HER2
- She told PharmaShots about how this test is able to identify patients who may benefit from Enhertu as a treatment option. She also elaborated on the key findings from the DESTINY-Breast04 trial under which the PATHWAY anti-HER2 (4B5) test was being evaluated.
- The approval of this test shows Roche’s vision to bring integrated, high-medical-value solutions, raising the standard of care
Shots:
- Nicolas discussed about Systemic Mastocytosis as a disease and highlighted its epidemiology & other details. He also spoke about the clinical data of avapritinib presented at the ASH’22 Annual Meeting
- Nicolas also shared the key findings from the PATHFINDER and EXPLORER trial evaluating avapritinib and the rationale behind conducting two different trials
- This interview gives an understanding of how Blueprint Medicines is developing life-changing therapies for people with cancer and blood disorders
Shots:
- Craig shared with our readers the preclinical study results of C101248 demonstrating its pharmacological activity and validation of the NETSseq platform as an effective method to interpret new therapeutic targets for Alzheimer’s disease
- He also elaborated on how the combination of the NETSseq platform and C101248 will help in the identification and development of novel therapeutics for neurodegenerative diseases
- The interview gives a glance at how Cerevance is advancing its pipeline of targeted treatments for patients with neurodegenerative diseases, including Alzheimer’s disease and Parkinson’s disease

Shots:
- The P-III trial (NETTER-2) evaluating Lutathera + long-acting octreotide in 222 patients with newly diagnosed somatostatin receptor (SSTR)-positive, grade 2 & 3, advanced gastroenteropancreatic neuroendocrine tumors (GEP-NETs)
- The trial met its 1EPs & demonstrated a significant improvement in PFS over high-dose long-acting octreotide alone while no new or unexpected safety findings were seen and the results were consistent with the well-established safety profile of Lutathera
- The results will be presented at an upcoming medical meeting and discussed with regulatory authorities. Lutathera, an advanced accelerator applications RLT was approved in the US for SSTR+ GEP-NETs
Kashiv Biosciences Reports P-I Clinical Study Results of ADL018 (biosimilar, omalizumab)
Shots:
- The P-I study has been completed evaluating ADL018, a biosimilar candidate to Xolair, EU-approved omalizumab, or US-licensed omalizumab via SC inj. in healthy volunteers
- The primary objective of the study was to demonstrate PK similarity by evaluating the area under the concentration-time curve from time zero to infinity (AUCinf) and Cmax and compare the safety & tolerability while the secondary objectives were to determine the PD activity & immunogenicity b/w three treatment groups
- The company plans to initiate the patients dosing in the P-III study shortly for CSU. Omalizumab was approved to treat persistent asthma in patients aged ≥6yrs. and chronic rhinosinusitis with nasal polyps in patients aged ≥18yrs.
Shots:
- The US FDA approves the expanded use of Antifungal Cresemba in the US in Children with Invasive aspergillosis (IA) & invasive mucormycosis (IM)
- The approval was granted based on the outcomes of two pediatric clinical studies, incl. a P-II study evaluating the safety, effectiveness, & PK of Cresemba in pediatric patients aged 1 to 17 years, for the treatment of IA and IM
- Additionally, the US FDA granted Cresemba pediatric exclusivity, extending U.S. market exclusivity to Sept 2027. In Europe, a similar application awaits EMA assessment, with a decision expected in H1’24. Cresemba is approved in 76 countries and marketed in 71, incl. the US & most EU states
Shots:
- SpliceBio will be eligible to receive up front, opt-in, and milestones of ~$216M along with royalties on net sales
- Spark to get an exclusive right globally to develop, manufacture, and commercialize a gene therapy arising from this research collaboration targeting an undisclosed inherited retinal disease
- The collaboration focuses on developing a gene therapy for an undisclosed inherited retinal disease using SpliceBio’s Protein Splicing platform. Additionally, Spark is working to create capabilities and a pipeline of wholly-owned gene therapy programs as well as to advance its lead program for the treatment of Stargardt disease
Shots:
- The P-II study evaluating Zynlonta (loncastuximab-tesirine-lpyl) + rituximab in patients (aged 47 to 89yrs) with r/r FL who are treated with ≥1 line of systemic therapy & meet GELF criteria or POD24 at baseline
- The 1EP of the study includes a complete response (CR) by week 12 on PET/CT. The result of the study depicted an ORR of 96.3% & CR rate of 85.2% along with a PFS of 92.3% with a median follow-up of 12mos. Additionally, the median PFAS was not met
- Zynlonta functions as ADC that targets CD19. When it binds to a cell expressing CD19, the cell internalizes it, & enzymes subsequently release a drug payload containing pyrrolobenzodiazepines (PBD)
Shots:
- The company initiates the P-II confirmatory patient study of AVT05, a biosimilar candidate to Simponi and Simponi Aria (golimumab) evaluating the efficacy, safety, and immunogenicity of AVT05 (SC) vs EU-approved Simponi in adult patients with mod. to sev. rheumatoid arthritis
- The primary outcome measures will be to show the comparative efficacy of AVT05 vs Simponi @16wks. based on change from baseline on a standardized DAS28-CRP which evaluates the severity of RA using clinical and laboratory data
- Additionally, the company initiates PK study for AVT05 in Jan 2023. Golimumab is a mAb that inhibits tumor necrosis factor-alpha (TNF alpha)
Shots:
- The US FDA has accepted to review the resubmitted NDA of SPN-830, apomorphine infusion device, for the treatment of motor fluctuations (OFF episodes) in Parkinson’s disease (PD) with the decision expected in Apr 2024
- The NDA was resubmitted following the initial refusal to file (RTF) due to inadequate data, which had occurred after the original NDA submission in Sep 2020
- The original NDA was based on data from the P-III (TOLEDO) trial that demonstrated a significant reduction in OFF time, with an improvement of 1.89hrs. per day vs PBO. These improvements were observed within 1wk. of initiating the therapy
Shots:
- The company highlighted the 1yr. results from the P-III study evaluating SB15 vs aflibercept. Additionally, 32wk. interim results from the same study were previously presented at AAO 2022
- SB15 showed comparable efficacy, safety, immunogenicity & PK profiles to reference aflibercept @~56wks. along with improvements in BCVA & anatomic outcomes. Switching from aflibercept to SB15 did not reveal any treatment-emergent issues i.e., loss of efficacy, increased AEs or immunogenicity,
- New analytical data on similarity b/w SB15 and the reference product were also presented where SB15 showed high similarity to AFL with respect to structural, physicochemical, and biological properties
Innate Pharma Reports Results from the P-II Study (TELLOMAK) of Lacutamab for Sézary Syndrome
Shots:
- The P-II (TELLOMAK) study investigated the safety and efficacy of lacutamab monotx. in patients (n=56) with r/r Sézary syndrome who had undergone at least two previous systemic therapies incl. mogamulizumab
- The study results, at the data cut-off of May 1, 2023, depicted that the global confirmed ORR was 37.5% incl. 2 CRs, confirmed ORR in skin was 46.4% incl. 5 CRs, confirmed ORR in blood was 48.2% incl. 15 CRs, CBR (CR+PR+SD) was 87.5% and m-PFS was 8.0mos. with a favorable safety profile
- The company will highlight these results along with the data from study of SAR443579/IPH6101 for r/r acute myeloid leukemia (AML), B-cell acute lymphoblastic leukemia (B-ALL) or high-risk myelodysplasia at ASH’23
Shots:
- The ongoing P-II study (OPTIMIZE-1) evaluating mitazalimab in combination with CT (mFOLFIRINOX) in patients with mPDAC. The results are expected in Q1’24
- The results showed an ORR of 52% in pre-planned interim efficacy analysis among 23 evaluable patients as per the response evaluation criteria in solid tumors (RECIST 1.1), DCR, ORR, or stabilization of disease was ≥90%
- Mitazalimab was safe and tolerable at 900µg/kg in combination with mFOLFIRINOX, as previously reported in the P-Ib dose escalation phase of the trial. The company intends to talk with regulators in the EU & EU about these positive results to explore a potential accelerated development and approval pathway for mitazalimab
Related Post: PharmaShots’ Most Read News of 2022